by Peter Vanovertveld & Günter Kloucek
There are seismic changes affecting the healthcare landscape. Disruptive technology, new competitors from outside the industry and increasing regulatory complexity are just a few of them. But the single most impactful is probably still a shift in in the balance of power in which Health Care Professionals (HCPs) can no longer decide on their own what therapies a patient will receive. In particular payers and patients are increasingly taking shared responsibility in this decision.
To create innovative solutions meeting the needs of all the stakeholders who influence patients access to medicines while taking account of the aforementioned shift in decision responsibility, an outside-in mindset and process that fosters collaboration is needed.
The Shifting Landscape
The voice of patients has gotten louder and more influential. Thanks to digital technology and access to their own health data, more and more patients are taking greater control of their own health. Digitalization is driving consumerization of health. The recent public health challenges around COVID have only reinforced this perception amongst the public that they have a say in choosing appropriate treatment for oneself.
As with consumer goods, patients expect the experience of health care services to be positive, tailored to their needs. Digitalization is also eroding national boundaries as the delivery of services and proximity of patient to HCP is increasingly less important e.g., app-based health tracking, management, and tele-medicine. Empowered patients, aware of what is available elsewhere, want to exercise their right to the best care possible. They are becoming empowered health care consumers.
Simultaneously, governments and payers are flexing their muscles, increasingly mandating what physicians are allowed to prescribe, demanding evidence that meets their standards for improved clinical and economic health outcomes. Evidence has become one of the most important building blocks for successful product launch.
For a science-based industry that is used to pushing products out into the market, these shifts in stakeholder balance of power call for a fundamental change in perspective to a holistic “outside-in” approach. As multiple stakeholders decide about patient access to medicine and its value, pharmaceutical companies have to start from the perspective of all stakeholders and follow a process to optimize value for all of them.
All Stakeholders Are Created Equal, Some More Than Others
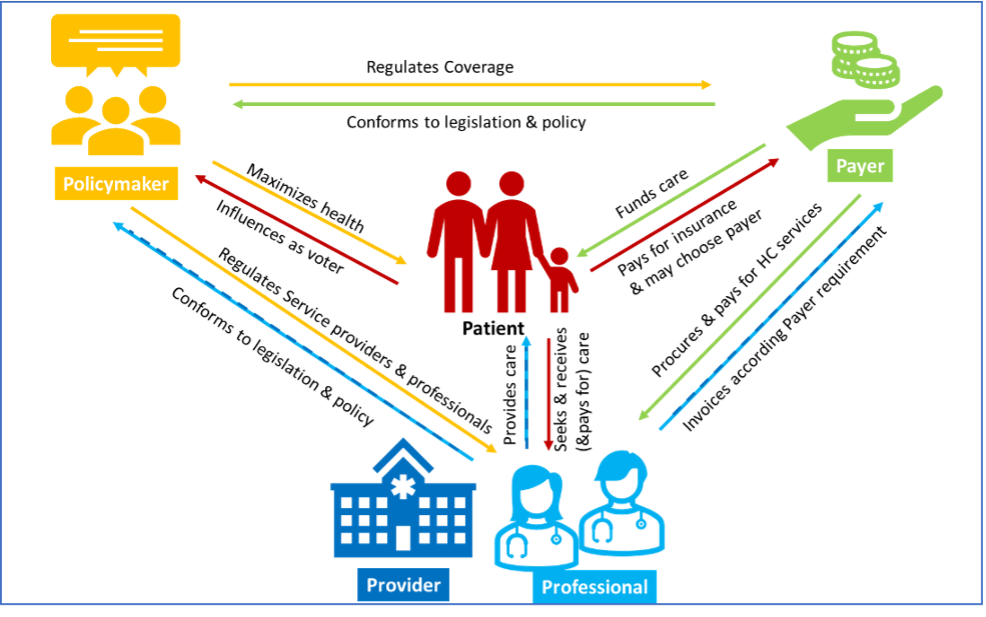
The concept of the 5Ps – patient, prescriber, provider, payer, policy maker – is a helpful approach to analyze the stakeholder perspectives and their relationship to each other:
- Policy makers establish the framework within which Health Care (HC) is provided to the country citizens and define relevant aspects such as size of investment in populations health, eligible care receivers, access to and funding of health services, health focus areas, etc. They regulate providers, prescribers and all healthcare professionals and payers with policies regarding what services can be provided how, where and by whom and how services are paid for. Policy Makers perspective: set the context within which the health care system operates and aim to maximize the health of the population within a country’s financial and resource constraints i.e., set the rules of the game.
- Payer is a body that finances or refunds cost of medicinal products and health services and operationalizes the financial aspects of the legislative framework. Payers are responsible for processing patient eligibility, for enrollment of beneficiaries, for procurement of high quality, cost effective services and for claims and payment management with service providers & professionals. Their willingness to pay for a health product or service may vary by geography, disease and perceived unmet medical need. Payers can be public or private; they are funded through insurance premium or tax/social insurance contributions. In some geographies patients may have a choice of payer aka insurer. Payer perspective: aim to address health needs of their beneficiaries while balancing conflicting goals of providing the state-of-the art therapies while treating their constituency equitably while minimizing costs i.e., decide on size of the prize.
- Provider is a body that operationalizes care delivery and administers health services. The term provider refers to institutional providers such as clinics, hospitals and provider networks. Within the policy framework, they create the organizational framework for prescribers and other care professionals to provide health services to patients, maintain health information about patients and coordinate care with other providers. Administrators are gatekeepers in provider organizations and exert powerful influence on formulary decisions, DRG implementation & optimization, negotiated rates and the adoption of new procedures. Provider organizations can be privately or publicly owned and fund themselves, depending on their legal structure, via tax payments, payments from payers and billing directly to patients. They can be for-profit and non-for-profit. Provider perspective: meet their obligations as a supplier in providing high quality care to patients in an effective & efficient way on behalf of their stakeholders (payers, public communities, patients) i.e., win for their stakeholders.
- Healthcare professionals (HCPs) including prescribers provide, within the legislative framework, health care treatment, service and advice to patients based on formal training and experience. HCPs are either employed at institutional providers or run independent businesses and must manage their own operations and finances. As independent businesses, professionals very often act as a supplier to payers. Embedded in provider or payer arrangements the nature of physician decision making has changed resulting in less clinical autonomy. Not at least as incentives for HCPs have changed,, besides financial incentives, clinical KPIs are increasingly added to traditional evaluations of patients’ improvements and well-being. Professional perspective: deliver high quality care to patients within policy framework and payer requirements while running own (or employer’s) business commercially successfully i.e., win while proving the best possible care for patients.
- Patients (citizens, taxpayers, voters) – receive care services from providers/professionals and are the beneficiary customer of the payors. They pay for health services directly or through taxes and social insurance contributions. As health seeker, the patient may have a choice of provider, professional and depending on the system also a choice of insurer. Patients have increasingly evolved from pure care recipients to healthcare consumers, willing and able to take greater control of their own health. Organized in patient advocacy groups, they are playing an increasingly important role in influencing health policy discussions. Patient perspective: increasingly are demanding tailored health solutions going beyond safety and efficacy, expect access to information and own health data via digital technologies and a consumer-like customer experience i.e., increasingly defining themselves what is good and what “winning” means to them.
The Balancing Act
The drivers of value for the different stakeholders vary. They have been and will continue to evolve over time. The perception of clinical and economic impact of a drug or a treatment is deeply rooted in the specific needs of a stakeholder thereby influencing their access decisions. An in-depth understanding of all stakeholders’ value drivers, unmet needs combined with a holistic view on their mutual dependencies and interactions is essential for the development of impactful value propositions and value-adding offerings.
- Evolution of thinking by regulators
Policy makers play an especially important role by shaping the rules and playing field when it comes to innovation including breakthrough therapies or digital health. Working with them on creating the right environment is key for significant uptake of the right innovation. In the past 20 years their thinking has evolved with the thinking of regulatory bodies. For example, from only accepting evidence from randomized and placebo controlled trials to the use surrogate markers and most recently to the acceptance of indirect comparisons and safety benefits using for instance synthetic control arms. Their methods of evaluation have evolved with evolving needs and technologies, especially ever improving means to capture high quality evidence, be that in a real-world or a trial setting.
- Opportunity for growth of payers
Most payers are still locked into traditional ways of defining value working with tools and assumptions like regulators were 20 years ago. They re-evaluate medicines after regulatory approval taking a look at the standard of care and evidence that new medicines provide additional value compared to products in use today. As for neglected or orphan diseases, what if there is no comparison product, yet the target population and unmet need are significant e.g., HIV in the 1980s? Payers have the opportunity to learn from regulators using more innovative methods for defining value that are needs based. However, although willingness, capability and infrastructure for innovative approaches such as value based pricing is growing in some geographies faster than others (e.g. Scandinavia), there is still a strong embrace of traditional health outcomes evaluations mainly driven by a short term cost-benefit analysis and discounting of long term benefits.
The perspective of an HCP differs from that of an (institutional) provider and from that of a patient when it comes to the “best” treatment alternative. For example, a patient may not choose the clinically most effective option, preferring one that they can best accommodate in their living situation e.g., not requiring adaptations at home. Weighing multiple views to get the balance right for the patient is the ultimate objective from Inside-Out to Outside-In.
Changing how we work
Historically, weighing and integrating the perspectives of all 5Ps is often impeded within pharmaceutical companies by how they are internally organized. Typically, they work in a siloed fashion with different functions responsible for addressing the need of a particular stakeholder:
- Regulators are the focus for Regulatory Affairs
- Medical & Scientific Community members are cared for by a company’s Medical Affairs professionals
- Payer relationships are managed by the Market Access function
- Prescribing physicians and care givers are the focus of marketing and sales
While there are signs of change out there, the prevailing perspective today is siloed and inside-out rather than holistic, outside-in, and patient focused. Specifically, what needs to shift is:
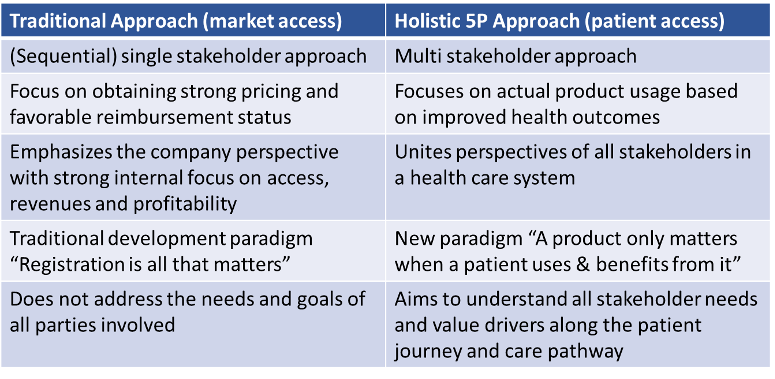
What is the best way to make the shift? The “patient at the center” is what the remaining 4Ps and Pharma have in common and the reason all the other stakeholders exist. Therefore, looking at the journey a patient experiences (from symptom to diagnosis to treatment) from all different stakeholder perspectives is the most effective way to gain the holistic view required for maximizing value generation, value demonstration and finally improved health outcomes. The patient journey provides both quantitative data (e.g. prevalent, diagnosed, treated patients) surrounding each milestone on the journey as well as qualitative data (e.g. patients and other stakeholders needs, thoughts and emotions along the way) and help identifying gaps in the care pathway. By taking a cross-functional, interdisciplinary view of this data, elevate relevant evidence on the patients’ experience, unmet needs are identified. And put simply, evidence of these unmet needs is the key to enhancing value.
Both a change in perspective, from inside-out to outside-in, and a process that is holistic and integrates perspectives and evidence are needed to maximize the total value of a new medicine or therapy.
P5 Process
To overcome the siloed ways of working in most pharmaceutical companies, a horizontal process that starts by collecting the unmet needs of all 5Ps and ends with insights and a feedback loop from patients as depicted below:
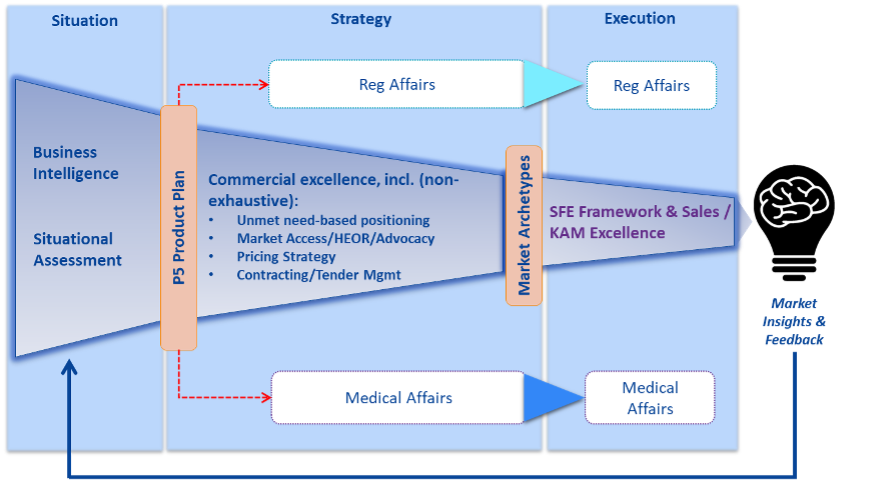
At the heart of the P5 process is the P5 plan that has the following elements:
- The unmet needs of all 5Ps
- The key issues that must be overcome to meet those needs
- Strategic initiatives to address the issues
- Concrete action plans
All of the above is considered in the context of the therapeutic area vision and desired product positioning. The issues are derived from a SWOT analysis relative to the ideal product positioning. It is at that point when each of the contributing functions can start to run in parallel, not before.
A holistic P5 approach encourages close collaboration of all functions along the entire value chain of the company during the life cycle of a product and processes and structures are in place to enable and foster this collaboration. It leads to breakthroughs such as medical affairs, patient advocacy and market access functions providing the view of their respective stakeholders early in the development phase to ensure use of holistic parameters for pipeline progress or clinical trial program decisions. Marketing and supply chain functions collaborate on solutions for patient-friendly distribution and delivery systems. Medical Affairs and Market Access collaborate in leveraging real world evidence (RWE) or patient reported outcomes (PRO) for value demonstration to achieve, improve and maintain access or develop pay-for-performance schemes. These and more concrete ways of collaborating are the natural result of using a P5 process.
Conclusion
The shifting landscape and changing balance of power from physicians to payers and patients necessitates a new mindset and holistic approach to maximizing value for all involved in order to provide patients with better access to medicines. There is no fault in acknowledging the 5Ps have distinct and sometimes competing objectives that can only be balanced when considered together. And their perspectives matter irrespective of market archetype.
A change in mindset is needed in most pharmaceutical companies from the traditional inside-out view to a holistic patient-centered, outside-in perspective based on sound evidence. A 5P process can overcome the tendency to work in functional siloes by first developing cross-functionally a 5P plan based on insights into the unmet needs of all stakeholders. Winning on behalf of the patients takes a team with the right mindset and gameplan.
References: INTRODUCING THE KEY STAKEHOLDERS: PATIENTS, PROVIDERS, PAYORS, AND POLICYMAKERS (THE FOUR P’S)
About the authors
Peter Vanovertveld is Chief Commercial Officer & Chief Operations Officer at Smart Reporting GmbH leading transformational change in healthcare towards personalized care through bottom-up and top-down efforts on better data input, evidence generation, and evidence usage. Previously Peter has acted as pharmaceutical executive holding various commercial leadership positions in several pharmaceutical companies for more than 15 years.
Günter Kloucek is a Director at OXYGY. Prior to consulting, Günter was the General Manager of Takeda Germany. He has more than 25 years of experience in the pharmaceutical industry and held operating company responsibility as well as led corporate functions including business strategy for the Europe & Canada region.
Acknowledgement
John Glasspool, the CEO and President of Anthos Therapeutics, is a pioneer of the P5 approach to whom we are grateful for his insights and leadership.