- Industry faces disruptive changes in the marketplace outdating the traditional business models
- The challenges are varied, complex and highly interdependent; therefore, a holistic and agile approach is required
- We recommend 6 principles to be considered for successful business model innovation to thrive in a landscape of outcome driven, personalized healthcare with empowered patients, fast advancing technology and increasing regulatory complexity
Introduction
The challenges life sciences companies face today are varied, complex and highly interdependent; therefore, call for a holistic approach to overcome them. The marketplace is being disrupted by a shift towards outcome-based funding, the empowerment of patients, disruptive technology, new competitors from outside the industry and increasing regulatory complexity. All of the above, mean the classic business model of life sciences companies is no longer adequate. There are six trends that present risks; but, also opportunities to innovate and lead the way forward.
Moving towards a health outcomes
Advances in medical science coupled with a growing and aging population increases the need for health resources globally. The rising demand for health care clashes with constrained budgets. Governments and payers are exerting pressure to drive down health care costs. Payers are realizing that driving down drug prices alone is not enough. Hence, they are demanding for improved health outcomes: clinical (reduction in morbidity and mortality) and/or economic (e.g. enhancing efficiency of care pathway, reducing resource consumption) and want to see strong real world evidence to support claims.
“Pharma will be paid for outcomes not for products in future.”
Although the willingness, capability and infrastructure for value-based pricing is growing faster in some countries than others, the trend is clear: increasingly pharma companies will be paid for outcomes, not for products. This move will consequently increase the pressure on pharma companies for value-based pricing, pay for performance and innovative solutions all backed by data to demonstrate value delivered.
Shift in stakeholder balance of power towards payers and patients
The need for demonstrating value based on health outcomes becomes even more pronounced as the balance of power shifts toward payers. Governments and payers are increasingly influencing what physicians are allowed to prescribe. Digital technology provides payers with outcome data they can use to determine best medical practice. Treatment protocols mandated by payers have started to replace individual treatment decisions by physicians.
Patients are becoming increasingly important stakeholders, willing and able (thanks to digital technology) to take greater control of their own health. Organized in patient advocacy groups, they are taking an increasingly important role in influencing health policy discussions. They want more than safety and efficacy. Patients are increasingly demanding tailored health solutions.
“Patients are becoming increasingly important stakeholders being able and willing to take greater control of their own health.”
For a science-based industry that is used to pushing products out into the market, the increased power of payers and patients calls for a 180o change in perspective to an “outside in” approach. Pharma companies have to start from the perspective of all stakeholders, including payers and patients, identify their unmet needs and value drivers. Based on those, develop new value propositions, service offerings and innovative ways of engaging to deliver greater value.
“Advances in digital technologies are a major contributor to the transformation of the life science industry.”
Digitalization is going to reshape and disrupt existing business models
Advances in digital technologies are a major contributor to the transformation of the life science industry. New health information technology provides real time access to health data and advanced analytics for processing an enormous amount of data from various sources, thereby enabling new forms of diagnostics, treatment and monitoring to personalize care. Digitalization is enabling the outcome driven health sector through technology for collection and analysis of data required to proof effectiveness in the real world. Digital technologies drive consumerization of healthcare as it empowers patients through increased access to information and health data. It opens up a range of independent information channels including online communities for sharing patient experience and apps for monitoring therapy impact.
Disruptive technologies and seminal therapies will significantly advance medical care …
A broad range of promising and disruptive technologies such as 3-D printing, nanotechnology, bionics and predictive analytics as well as seminal new treatments like genetics, cellular programming and stem cell therapies are starting to have an impact on today’s medical care. Those advances are going to significantly enhance patient care shifting from treatment of symptoms to predictive diagnostic, prevention and cure. Overall, healthcare delivery is becoming more tailored to individual patient needs whether on a molecular level or through specific disease management services.
… and attract new players
“This development is going to attract disruptive new entrants with unique digital and patient engagement capabilities.”
This development is going to attract new entrants from within and outside the industry. The “who’s who” of technology are moving into healthcare with huge investments, agility and unique capabilities in data collection and processing as well as in health consumer engagement: companies like Apple, Google, IBM, Amazon and Microsoft. Those new entrants are disrupting the marketplace and changing the role of existing players in the health care continuum potentially making the traditional pharma business model obsolete, replacing it with innovative models based on data, analytics and end consumer relationships.
Shaping and navigating regulations is a mission critical capability
The already complex regulatory landscape is going to become even more complicated as the demand for health care solutions increases. When it comes to data privacy, nothing is more sensitive than personal health data. Add to that the need for new regulations on related topics: new technology standards, new scientific standards for RWE, security regulations and transparency rules to name a few. Emerging regulations will define boundaries within which the actors in the health care sector are allowed to play. Now is the time for pharma companies to bring their expertise to appropriately shape regulatory guidelines for the benefit of all stakeholders. At a minimum, the capability to navigate in a compliant way an uncertain and ambiguous regulatory landscape successfully has become a business critical capability.
The call for business model innovation is loud and clear
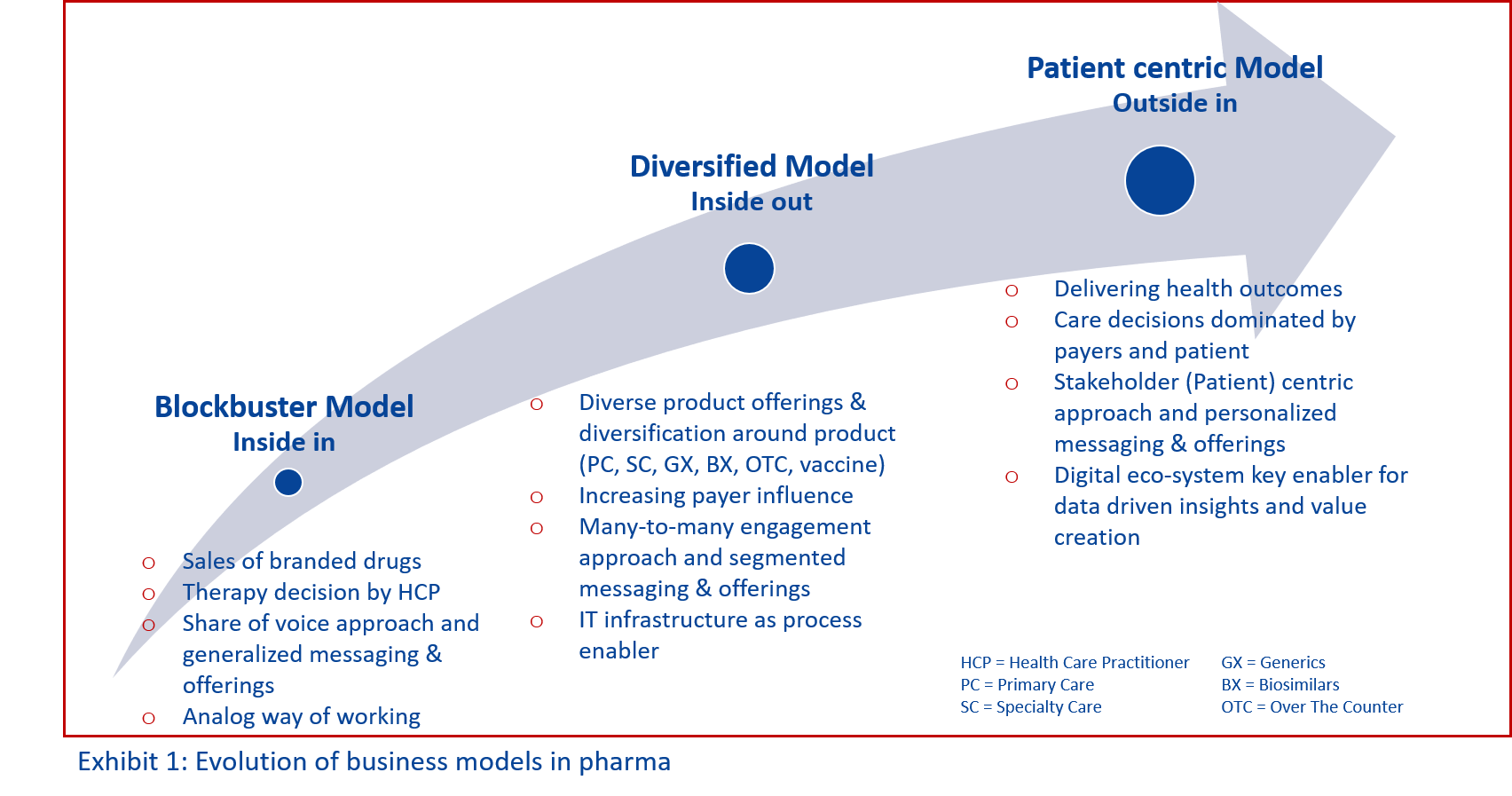
These key trends underpin what is happening across the life sciences sector: the era of the traditional blockbuster approach to business is over. The ability to develop promising molecules and push branded products with substantial sales and marketing efforts into markets is no longer sustainable. It no longer meets changing market requirements. There are better ways that avoid the dramatic loss in revenue every 10 years that comes with the loss of exclusivity of blockbuster drugs.
To thrive in a landscape of outcome driven, personalized healthcare with empowered patients and fast advances in technology a significant change in business models is inevitable. (see Exhibit 1)
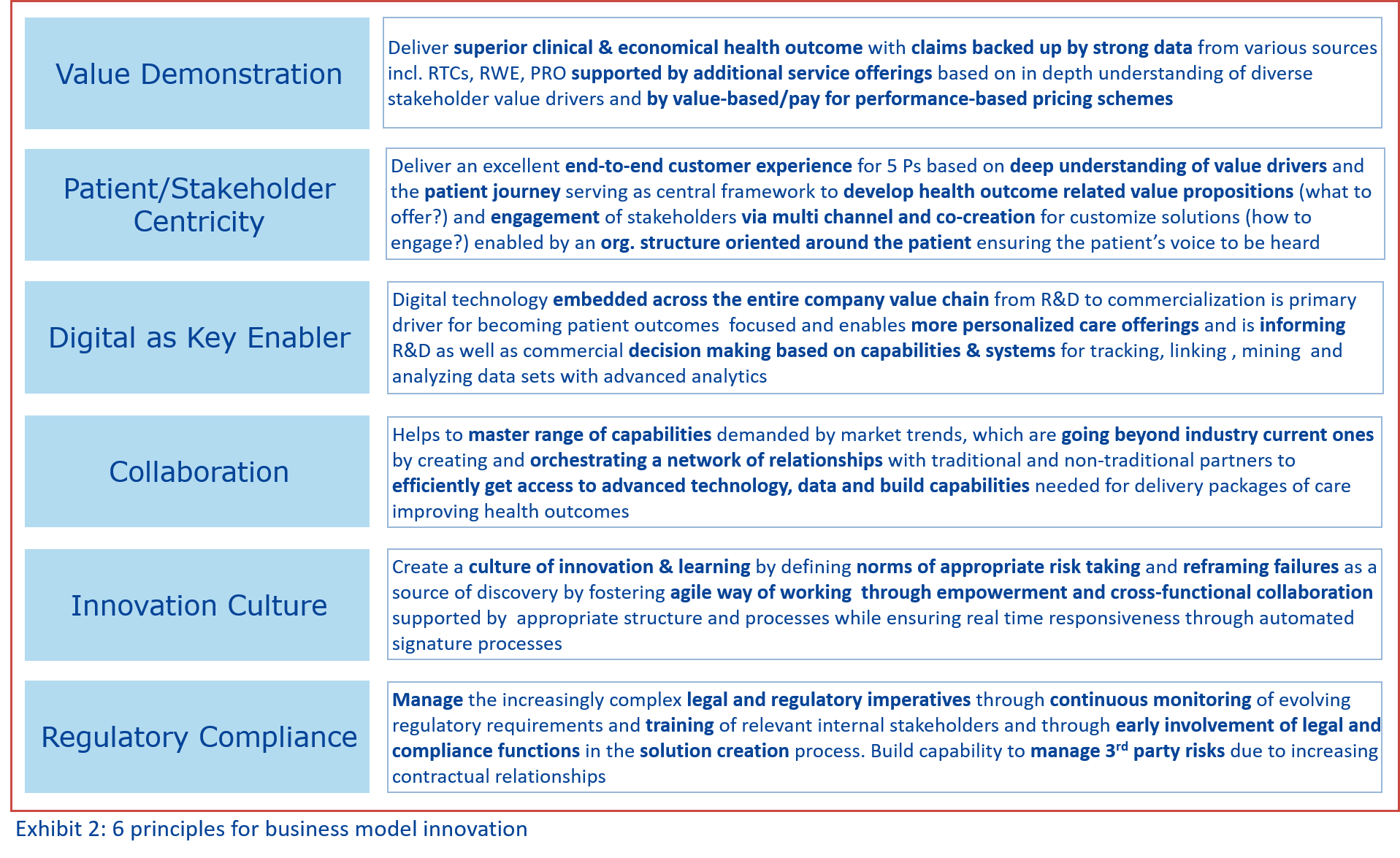
Irrespective of legal structure, organization form or portfolio focus, we believe that the following 6 principles are vital and need to be considered in adapting or building a new business model. (see Exhibit 2)
1. Value Demonstration
“The value through the drug needs to be complemented by the value beyond the drug.”
In an outcome driven healthcare system, the ability to demonstrate the value of your offering is key. Classical clinical trials, although important from a regulatory approval perspective, have their limitations. Real World Evidence (RWE) and Patient Reported Outcomes (PRO), complementing data from clinical trials and showing effectiveness of a medication in practice, can serve as basis for pay for performance and demonstration of “real” value for money. The collection of RWE from diverse appropriate sources such as registries, patient health apps or even social media requires building strong RWE capabilities and reliable infrastructures. However, value demonstration for a drug itself can, despite strong clinical product data, become increasingly difficult since many factors such as adherence and insufficiencies in care provision influence outcomes. To create a compelling value demonstration, managing the transformation from “pure drug provision” into the health management space becomes vital. Based on an in-depth understanding of value drivers of different stakeholders the provision of additional services and solutions around the drug intended to improve health outcomes by personalization and tailoring of care will become a key feature to demonstrate value.
2. Patient/Stakeholder Centricity
“Becoming a preferred partner to diverse stakeholder means delivering an excellent end-to-end customer experience”
Developing a stakeholder centric model encompassing this shift in power becomes an imperative. The varying needs and value drivers of the 5 P’s (physician, provider, policy maker, payer and patient) need to be fully understood. The needs of patients gives the other stakeholders the right to exist. Therefore, the patient journey must serve as the central framework for developing activities, health outcome related value propositions and engagement models. To become truly patient centric requires moving from a paternalistic, transactional to a transformational approach to personalized care. Patient interests have to be put in the center of a company‘s decision making process and actions, aiming for improved patient experience and patient value creation (from the patient’s perspective, of course). Engaging patients in the co-creation of health solutions across the entire value chain all the way from research & development (R&D) to commercialization is the best way to ensure that pharma companies are truly focused on delivering greater value to patients.
3. Digital Enables Value Creation
Recent surveys show many life science companies are still in the exploration phase when it comes to the adoption of digital technology (Accenture 2016, McKinsey 2016, Capgemini 2018, Across Health 2019). Embedding health technologies along the entire value chain of an organization has the potential to deliver dramatic benefits:
- enhance R&D productivity
- demonstrate greater value
- deliver personalized care
- enrich stakeholder engagement via multiple channels
- automate business processes; and,
- improve decision making based use data driven insights.
For all the above reasons, digital technology has to be an integral part of any new business model in life sciences. However, the benefits will only be realized when the importance of digital and its impact on the way of working is fully understood. It is having an culture of innovation, not just in science but ways of working, that will separate those who can take advantage of this opportunity from those whose risk averse and siloed ways of thinking will hold them back.
4. Collaboration and Partnership
To take advantage of these trends in life science successfully, the industry needs a far wider range of skills that go beyond its current capabilities. Building those required capabilities on its own is certainly a costly and time-consuming exercise and one of the paradigms in the industry “We better do it on our own to maintain control.” The future sector leaders will be faster and more cost effective by partnering and collaborating, with traditional stakeholders (academic institutions, patient advocacy groups, payers) and non-traditional partners such as technology, digital platform providers, biotech start-ups that give them access to technology, data and new capabilities required for innovative business models. Partnering and collaboration may be the only way to deliver packages of care improving health outcomes for which society is willing to pay in future.
5. Innovation Culture
We believe a prerequisite for designing and implementing new business models is a culture of learning and innovation. In order to move from rigid and risk averse to agile, experimental and adaptive behaviors, companies need to let go of a prevailing mindset: an idea has to be fully baked and a solution has to have every “bell & whistle” before being launched. Instead organizations need to foster an agile way of working and empower employees. A trust and learn mentality will reframe mistakes as a source of discoveries, reducing the fear of failure. Norms for appropriate risk taking will be defined. Creating agile organizations and developing agile leaders is the foundation for bring new innovative business models to life (Crom 2020).
6. Regulatory Compliance
It is a paradoxical world: while regulatory compliance is increasing in complexity, more agility and appropriate risk-taking behavior is required to manage the market challenges. If not done properly, designing a simple patient engagement activity like a disease support app will run into compliance issues: patient promotion-, data privacy-, pharmacovigilance-regulations, anticorruption and competition law, just to name a few. As interdependence between the players in the industry increases, company’s who are seen as ethical and trust worthy will be the partners of choice, especially for regulatory authorities. Involve legal and compliance functions early in the iterative solution creation process to ensure compliance and at the same time allow for an agile way of working. Companies with the ability to shape and manage the complex legal and regulatory imperatives will increasingly have a competitive advantage.
A holistic and agile transformation approach is required
Building new business models to take advantage of strategic trends, encompassing the 6 principles calls for a holistic and agile transformation approach.
What next steps should companies take:
- Understand your own maturity: How fit for the future are you? (click to download)
- Envision your future business model: What is the desired state?
- Align on priorities based on your business strategy and identified gaps: Where should we focus our efforts to achieve our ambition?
- Scope, charter and execute initiatives with an agile approach
- Monitor progress and continuously re-assess maturity (back to #1) and adapt approach as required
Conclusion
Today’s world of rapid changes in the life science landscape brings challenges for the industry but opportunities likewise. To thrive in the new market reality, leadership teams need to (re)articulate their purpose, envision their future business model and plot the path towards the desired state, thereby considering the type of technology, structure and processes, capabilities and job-profiles, culture and leadership model needed while engaging the organization in the transformation journey in an agile way.
Many thanks to Steve Crom and Arne Buthmann of OXYGY as contributors.
Free Download: Self-Assessment (Excel for Windows only)
Assess the maturity of your company regarding the 6 principles of innovative business models. Use the free download of our self-assessment tool. Please contact us if you need any support downloading or launching the tool. I would be happy to discuss the results with you in detail. Just write an email to: guenter.kloucek@oxygyconsulting.com.
Click here to download the Self-Assessment
About the author
Günter Kloucek is a Director at OXYGY. Prior to consulting, Günter was the General Manager of Takeda Germany. He has more than 25 years of experience in the pharmaceutical industry and held operating company responsibility as well as led corporate functions including business strategy for the Europe & Canada region.
References
Accenture; Industry at Crossroads: The rise of Digital in the Outcome Driven R&D Organization, 2016
McKinsey; Closing the digital gap in pharma, Nov. 2016
Capgemini; Digital Patient Engagement; Mar. 2018
Across Health; 10th annual Multichannel Maturometer Study on Digital Maturity; Apr. 2019